|
The group 16 elements each add two electrons, while the group 17 elements add one electron per atom, to form the anions. Notice that the group 16 ions are larger than the group 17 ions. 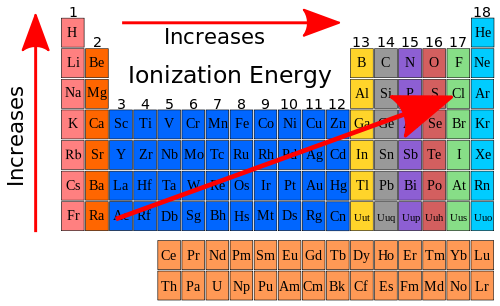
The electron cloud also spreads out because more electrons results in greater electron-electron repulsion. When the electrons outnumber the protons, the overall attractive force that the protons have for the electrons is decreased. This allows for a predictable way for electrons to fill up an atom and helps explain why elements in the same group on the periodic table have similar chemical. The addition of electrons always results in an anion that is larger than the parent atom. Shells furthest from the nucleus have the highest amounts of energy and those nearest have the lowest. The potassium atom has one electron removed to form the corresponding ion, while calcium loses two electrons. The shells exist at different levels around the nucleus. One other factor is the number of electrons removed. Scientists use the periodic table to quickly refer to information about an element, like atomic mass. Another reason is that the remaining electrons are drawn closer to the nucleus because the protons now outnumber the electrons. The periodic table of chemical elements, often called the periodic table, organizes all discovered chemical elements in rows (called periods) and columns (called groups) according to increasing atomic number. The periodic table is the arrangement of elements in rows and columns according to atomic numbers. When the valence electron(s) are removed, the resulting ion has one fewer occupied principal energy level, so the electron cloud that remains is smaller. Electrons always go into the lowest energy level first, until that energy level is filled. Group 2 elements represent the second column of elements on the left side of the periodic table. Columns are called groups, and the group number equals the number of electrons an atom of that element has in its outer shell. The removal of electrons always results in a cation that is considerably smaller than the parent atom. In the periodic table, the alkaline earth metals group is found in group 2. (Credit: Zachary Wilson Source: CK-12 Foundation License: CC BY-NC 3.0(opens in new window)) Groups 16 and 17 are nonmetals and form anions, shown in purple. Specifies the energy of an electron and the size of the orbital (the distance from the nucleus of the peak in a radial probability distribution plot). Principle energy levels are color coded, while sublevels are grouped together, and each circle represents an orbital capable of holding two electrons. Only carbon has the s 2 outer configuration, which accounts for some of the differences between carbon and other elements in the family. In other words it represents the change in energy occurring when an electron is. Two of these electrons are in the s subshell, while 2 are in the p subshell. Finally, electron affinity refers to an atoms ability to accept electrons. Groups 1, 2, and 13 are metals and form cations, shown in green. Carbon family elements contain atoms that have 4 electrons in their outer energy level. However, these elements are reactive enough that they do not exist in their elemental forms in nature, but are present as compounds.\): Comparison of ion sizes for Groups 1, 2, 13, 16, and 17. The need to remove two electrons in order for the material to react means more energy is needed for electron removal. The Group 2 elements tend to be less reactive than their Group 1 counterparts. :max_bytes(150000):strip_icc()/periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
However, radium is a radioactive element and is generally under the category of radioisotopes in addition to being an alkaline earth metal, because it is not a stable element. We can write the configuration of oxygens valence electrons as 2s☢p. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. Elements in the same group have the same number of outer electrons and have. Let's look at the elements sodium (Na), aluminum (Al) and potassium (K). Valence electrons are the electrons in the outermost shell, or energy level, of an atom. The period number (n) is the outer energy level that is occupied by electrons. Radium (atomic number 88) has similar properties to barium and is also in the Group 2 category. The numbers in front of the s, p, d, and f blocks on the periodic table signify the energy levels. \) (Credit: Ingmar Runge Source: (opens in new window) License: Public Domain)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
