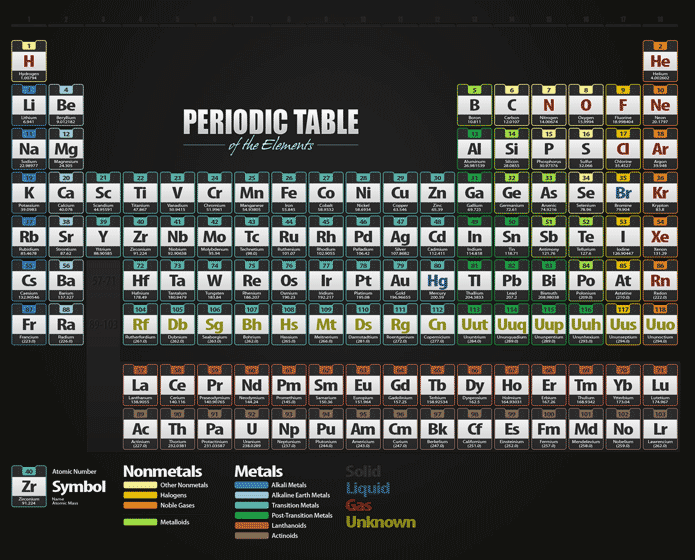
Unfortunately, there was a slightly different system in place in Europe. The first two groups are 1A and 2A, while the last six groups are 3A through 8A. The traditional system used in the United States involves the use of the letters A and B. There are two different numbering systems that are commonly used to designate groups, and you should be familiar with both. These two rows are pulled out in order to make the table itself fit more easily onto a single page.Ī group is a vertical column of the periodic table, based on the organization of the outer shell electrons. Periods 6 and 7 have 32 elements, because the two bottom rows that are separate from the rest of the table belong to those periods. Some of the groups have widely used common names, including the alkali metals (group 1. Elements that exhibit similar chemistry appear in vertical columns called groups (numbered 118 from left to right) the seven horizontal rows are called periods. Period 1 has only two elements (hydrogen and helium), while periods 2 and 3 have 8 elements. The periodic table is an arrangement of the elements in order of increasing atomic number. Both groups and periods reflect the organization of electrons in atoms. A new period begins when a new principal energy level begins filling with electrons. A group is a vertical column down the periodic table, while a period is a horizontal row across the table. There are seven periods in the periodic table, with each one beginning at the far left. The Arabic numbering system is the most widely accepted today.\) (Credit: User:Cepheus/Wikimedia Commons Source: (opens in new window) License: Public Domain)Ī period is a horizontal row of the periodic table. Many periodic tables include both Roman and Arabic numbers. The modern IUPAC system uses Arabic numbers 1-18, simply numbering the columns of the periodic table from left to right.The CAS system used letters to differentiate main group (A) and transition (B) elements.The basic metals make up the element to the right of the transition metals. The transition elements, groups IB to VIIIB, are also considered metals. The older IUPAC system used Roman numerals together with letters to distinguish between the left (A) and right (B) side of the periodic table. Group IA and Group IIA (the alkali metals) are the most active metals.Three systems have been used to number families and groups: Recognizing Families on the Periodic TableĬolumns of the periodic table typically mark groups or families. All elements in the alkali metal group occur in nature. Alkali metals are very reactive chemical species that readily lose their one valence electron to form ionic compounds with nonmetals. Alkali metals are any of the elements found in Group IA of the periodic table (the first column). Noble Gases: - Group 18 (VIIIA) - 8 valence electrons Chemistry Glossary Definition of Alkali Metal.Halogens: - Group 17 (VIIA) - 7 valence electrons. 
Oxygen Group or Chalcogens: - Group 16 (VIA) - 6 valence electrons.Groups: The vertical column of the periodic table that signifies the number. Nitrogen Group or Pnictogens: - Group 15 (VA) - 5 valence electrons Elements: A pure substance composed of a single atom with a unique atomic number. 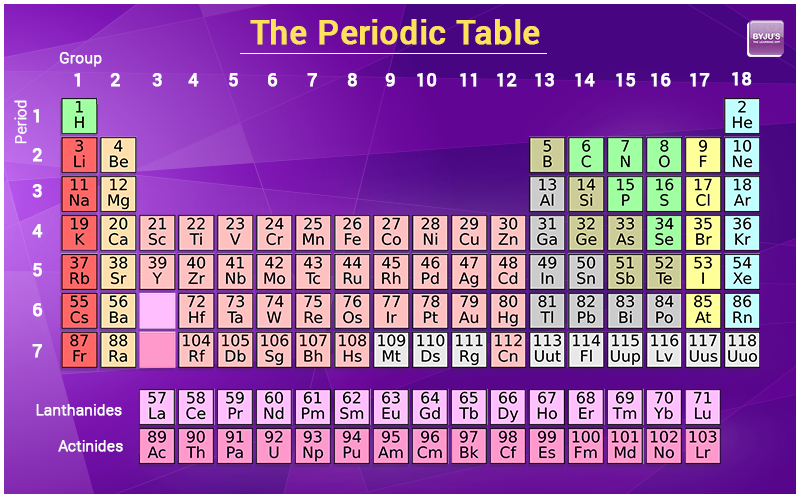
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
